 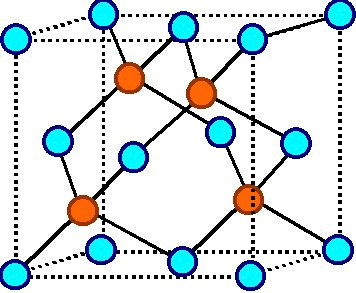
Most macroscopic inorganic solids are polycrystalline, including almost all metals, ceramics, ice, rocks, etc. In the final block of ice, each of the small crystals (called " crystallites" or "grains") is a true crystal with a periodic arrangement of atoms, but the whole polycrystal does not have a periodic arrangement of atoms, because the periodic pattern is broken at the grain boundaries. For example, when liquid water starts freezing, the phase change begins with small ice crystals that grow until they fuse, forming a polycrystalline structure. ( Quasicrystals are an exception, see below). A crystal is a solid where the atoms form a periodic arrangement. The scientific definition of a "crystal" is based on the microscopic arrangement of atoms inside it, called the crystal structure. amorphous solids.Ĭrystals, or crystalline solids, are often used in pseudoscientific practices such as crystal therapy, and, along with gemstones, are sometimes associated with spellwork in Wiccan beliefs and related religious movements. Examples of amorphous solids include glass, wax, and many plastics.ĭespite the name, lead crystal, crystal glass, and related products are not crystals, but rather types of glass, i.e. A third category of solids is amorphous solids, where the atoms have no periodic structure whatsoever. 
Polycrystals include most metals, rocks, ceramics, and ice. many microscopic crystals fused together into a single solid. Most inorganic solids are not crystals but polycrystals, i.e. Įxamples of large crystals include snowflakes, diamonds, and table salt. The word crystal derives from the Ancient Greek word κρύσταλλος ( krustallos), meaning both " ice" and " rock crystal", from κρύος ( kruos), "icy cold, frost". The process of crystal formation via mechanisms of crystal growth is called crystallization or solidification. The scientific study of crystals and crystal formation is known as crystallography. In addition, macroscopic single crystals are usually identifiable by their geometrical shape, consisting of flat faces with specific, characteristic orientations. Some solids, however, are intrinsically amorphous, because either their components cannot fit together well enough to form a stable crystalline lattice or they contain impurities that disrupt the lattice.Microscopically, a single crystal has atoms in a near-perfect periodic arrangement a polycrystal is composed of many microscopic crystals (called " crystallites" or "grains") and an amorphous solid (such as glass) has no periodic arrangement even microscopically.Ī crystal or crystalline solid is a solid material whose constituents (such as atoms, molecules, or ions) are arranged in a highly ordered microscopic structure, forming a crystal lattice that extends in all directions. Almost any substance can solidify in amorphous form if the liquid phase is cooled rapidly enough. An amorphous, translucent solid is called a glass. 
When cleaved or broken, they produce fragments with irregular, often curved surfaces and they have poorly defined patterns when exposed to x-rays because their components are not arranged in a regular array. Thus the intermolecular forces holding the solid together are uniform, and the same amount of thermal energy is needed to break every interaction simultaneously.Īmorphous solids have two characteristic properties. The resulting repulsive interactions between ions with like charges cause the layers to separate.Ĭrystals tend to have relatively sharp, well-defined melting points because all the component atoms, molecules, or ions are the same distance from the same number and type of neighbors that is, the regularity of the crystalline lattice creates local environments that are the same. Deformation of the ionic crystal causes one plane of atoms to slide along another. \): Cleaving a Crystal of an Ionic Compound along a Plane of Ions. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
